(This is a draft and truncated version - for final and full version, see
Concise Encyclopedia of Biostatistics for Medical Professionals)
crossover designs/ trials, see also crossover trials (analysis of)
A design is called crossover when the same groups of subjects are administered two or more regimens one after the other in one sequence in one group and in another sequence in the other group. In a simple 2×2 situation, this means that one group of subjects receives AB sequence (regimen-A followed by regimen-B) and the other group receives BA sequence. Since the subjects are the same in each group, this design helps to control between subjects variation that would occur if the groups of subjects are different and thus can provide substantially more believable results when the stringent conditions under which it is applicable are fulfilled. Crossover design has pronounced applications to clinical as well as laboratory experiments. Thus, the following discussion is mixed for laboratory and clinic setups.
Medicine is an incomplete science as it is today; many drugs work for a limited period. They have to be administered again, else the disease returns with the same intensity. Some drugs do have some carry-over effect but in others there is practically none. When all traces of their effect vanish, say two or three days after the last ingestion, the same subject can be used again to try the other drug, provided the condition of the subject reverses to what it was. This is called the steady-state. In the case of humans, epilepsy, migraine, enlarged prostate, and end-stage renal failure are examples of such diseases. In some patients, hypertension and diabetes require daily medication, otherwise the disease returns to the baseline. Yet, a high-cholesterol diet consumed by older subjects for a long time when they were young because of lack of awareness might still have a carry-over effect that persists despite a change to a low-cholesterol diet. Cross-sectional studies of the type envisaged by crossover design fail to take care of such confounders.
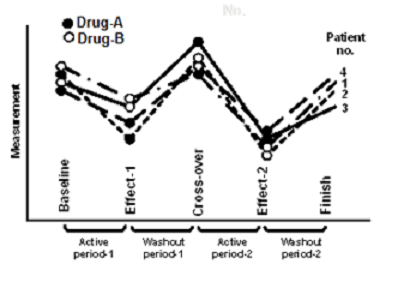
In the case of animals, the experiment could be for the effect of cocaine versus saline on behavior in terms of distance traveled in 30 min following the injection separately in long-sleep and short-sleep mice. In this experiment, some mice may receive cocaine on day-1 and saline on day-2 and others saline on day-1 and cocaine on day-2. The process of trying two regimens on the same subject after providing a time gap (called washout period) for complete disappearance of the carry-over effect of the first regimen is called crossover (Figure C.30).
Figure C.30: A representation of crossover design
You can see that crossover is more suitable for quantitative outcomes rather than binary (yes/no) response. Nonetheless, it can be used for such binary response as well. Suitability of crossover also depends on the regimen. Quickly reversible regimens are obviously more suitable than long-acting regimen such as steroids. Crossovers are difficult to implement for multidose trials.
An important prerequisite of a successful crossover experiment is the absence of carry-over effect. The carry-over effect need not be just physical but can also be psychological if that affects pharmacological outcome. Also, a drug may have caused damage to the liver that would affect metabolization of future drugs for a long time. If a patient receives regime-A followed by regimen-B, the effect seen after regimen-B would be the direct effect of regimen-B plus residual effect of regimen-A received earlier. This produces bias. The problem confounds further if the carry-over effect of regimen-A is different from the carry-over effect of regimen-B.
As just mentioned, the time gap required for carry-over effect to vanish is called washout period. This generally should be at least four times the half-life of the regimen. Half-life is the period at which half of the peak concentration of drug is available in the body and the other half is eliminated or metabolized by the body after the time when the peck is reached. Nothing should occur during the washout period that can affect the outcome. Absence of carry-over effect can be statistically tested in a crossover design. If carry-over effect is significant, think of an alternative such as Balaam design where AA and BB sequences are also included in addition to AB and BA sequence.
In a crossover experiment, the subjects are divided into two equal groups by randomization—one group receives drug-A followed by drug-B and the other receives drug-B followed by drug-A. Allocation can be done by tossing a coin or by any other random method. It is desirable to continue observation till the end of the washout period of the second drug. This provides confidence regarding the drug exiting the system. However, this sequence itself can cause differential effect. This can particularly happen when the intervening period is favorable to one regimen than the other. For example, when lisinopril is given first and then losartan after a 2 week washout gap to nondiabetic hypertensive patients for insulin sensitivity, the first drug may still be in the system. This may not happen when losartan is given first. Crossover design is not appropriate for such regimens that have significant sequence effects
The advantage of crossover is the availability of natural controls because the same experimental unit is used twice. This tends to reduce the impact of experimental error. The benefit of randomization is also available. Fewer subjects are required in this design relative to a parallel design with a separate control group. It is not necessary to recruit all subjects at a particular point of time. Period has a relative meaning. Some patients may have completed both periods before others are even recruited.
There are many cautions for using crossover design. Besides the basic requirement of no carry-over effect, crossover is recommended when the effect of the intervention can be assessed quickly so that the patient can be crossed over without a large time gap to deny opportunity for confounders to appear. If baseline condition can affect the outcome, all the subjects in the trial should be homogenous. Crossover is not a suitable design for cyclic diseases such as asthma and arthritis because they have natural remission periods. As for any design, there should not be any concomitant medication or comorbidity that can affect the outcome. Note that in this design, the subjects must stay in the experiment twice as long as otherwise needed in an experiment with parallel control group. This can cause higher dropouts. Crossover should not be used when substantial dropout is anticipated because then the statistical analysis becomes complex and the reliability steeply declines. However, periods in the case of crossover are not necessarily calendar periods. It is possible that one subject receives first-period treatment in the month of June and second period in the month of July, and the second subject is recruited in August for the first-period treatment. Also, the entire trial should not be modified mid-way in any manner such as for dosage, period, and sequence, because that could complicate the analysis.
Crossover is relatively easy when the treatment can be rapidly started and stopped. However, crossover strategy with random allocation of the order of therapy and adequate washout period as discussed earlier is essential for valid results from such self-control designs. Crossover design cannot be adopted if cure or death is the outcome. In addition, regulatory agencies seldom approve crossover because of the possibility of carry-over effect even if demonstrated to be absent. Also, side-effects and tolerability are difficult to study in a crossover setup since the administration is for a short period. Thus, crossover is not recommended for phase-III trials. But this design can be easily used in phase-II, particularly for identifying the right dosage. The conventional crossover is two regimens–two periods–two sequences strategy. But it can be extended for multiple periods and multiple regimens. For further details, see Senn [1]. ... ...
For final and full version, see
Concise Encyclopedia of Biostatistics for Medical Professionals
Concise Encyclopedia of Biostatistics for Medical Professionals)
crossover designs/ trials, see also crossover trials (analysis of)
A design is called crossover when the same groups of subjects are administered two or more regimens one after the other in one sequence in one group and in another sequence in the other group. In a simple 2×2 situation, this means that one group of subjects receives AB sequence (regimen-A followed by regimen-B) and the other group receives BA sequence. Since the subjects are the same in each group, this design helps to control between subjects variation that would occur if the groups of subjects are different and thus can provide substantially more believable results when the stringent conditions under which it is applicable are fulfilled. Crossover design has pronounced applications to clinical as well as laboratory experiments. Thus, the following discussion is mixed for laboratory and clinic setups.
Medicine is an incomplete science as it is today; many drugs work for a limited period. They have to be administered again, else the disease returns with the same intensity. Some drugs do have some carry-over effect but in others there is practically none. When all traces of their effect vanish, say two or three days after the last ingestion, the same subject can be used again to try the other drug, provided the condition of the subject reverses to what it was. This is called the steady-state. In the case of humans, epilepsy, migraine, enlarged prostate, and end-stage renal failure are examples of such diseases. In some patients, hypertension and diabetes require daily medication, otherwise the disease returns to the baseline. Yet, a high-cholesterol diet consumed by older subjects for a long time when they were young because of lack of awareness might still have a carry-over effect that persists despite a change to a low-cholesterol diet. Cross-sectional studies of the type envisaged by crossover design fail to take care of such confounders.
In the case of animals, the experiment could be for the effect of cocaine versus saline on behavior in terms of distance traveled in 30 min following the injection separately in long-sleep and short-sleep mice. In this experiment, some mice may receive cocaine on day-1 and saline on day-2 and others saline on day-1 and cocaine on day-2. The process of trying two regimens on the same subject after providing a time gap (called washout period) for complete disappearance of the carry-over effect of the first regimen is called crossover (Figure C.30).
Figure C.30: A representation of crossover design
You can see that crossover is more suitable for quantitative outcomes rather than binary (yes/no) response. Nonetheless, it can be used for such binary response as well. Suitability of crossover also depends on the regimen. Quickly reversible regimens are obviously more suitable than long-acting regimen such as steroids. Crossovers are difficult to implement for multidose trials.
An important prerequisite of a successful crossover experiment is the absence of carry-over effect. The carry-over effect need not be just physical but can also be psychological if that affects pharmacological outcome. Also, a drug may have caused damage to the liver that would affect metabolization of future drugs for a long time. If a patient receives regime-A followed by regimen-B, the effect seen after regimen-B would be the direct effect of regimen-B plus residual effect of regimen-A received earlier. This produces bias. The problem confounds further if the carry-over effect of regimen-A is different from the carry-over effect of regimen-B.
As just mentioned, the time gap required for carry-over effect to vanish is called washout period. This generally should be at least four times the half-life of the regimen. Half-life is the period at which half of the peak concentration of drug is available in the body and the other half is eliminated or metabolized by the body after the time when the peck is reached. Nothing should occur during the washout period that can affect the outcome. Absence of carry-over effect can be statistically tested in a crossover design. If carry-over effect is significant, think of an alternative such as Balaam design where AA and BB sequences are also included in addition to AB and BA sequence.
In a crossover experiment, the subjects are divided into two equal groups by randomization—one group receives drug-A followed by drug-B and the other receives drug-B followed by drug-A. Allocation can be done by tossing a coin or by any other random method. It is desirable to continue observation till the end of the washout period of the second drug. This provides confidence regarding the drug exiting the system. However, this sequence itself can cause differential effect. This can particularly happen when the intervening period is favorable to one regimen than the other. For example, when lisinopril is given first and then losartan after a 2 week washout gap to nondiabetic hypertensive patients for insulin sensitivity, the first drug may still be in the system. This may not happen when losartan is given first. Crossover design is not appropriate for such regimens that have significant sequence effects
The advantage of crossover is the availability of natural controls because the same experimental unit is used twice. This tends to reduce the impact of experimental error. The benefit of randomization is also available. Fewer subjects are required in this design relative to a parallel design with a separate control group. It is not necessary to recruit all subjects at a particular point of time. Period has a relative meaning. Some patients may have completed both periods before others are even recruited.
There are many cautions for using crossover design. Besides the basic requirement of no carry-over effect, crossover is recommended when the effect of the intervention can be assessed quickly so that the patient can be crossed over without a large time gap to deny opportunity for confounders to appear. If baseline condition can affect the outcome, all the subjects in the trial should be homogenous. Crossover is not a suitable design for cyclic diseases such as asthma and arthritis because they have natural remission periods. As for any design, there should not be any concomitant medication or comorbidity that can affect the outcome. Note that in this design, the subjects must stay in the experiment twice as long as otherwise needed in an experiment with parallel control group. This can cause higher dropouts. Crossover should not be used when substantial dropout is anticipated because then the statistical analysis becomes complex and the reliability steeply declines. However, periods in the case of crossover are not necessarily calendar periods. It is possible that one subject receives first-period treatment in the month of June and second period in the month of July, and the second subject is recruited in August for the first-period treatment. Also, the entire trial should not be modified mid-way in any manner such as for dosage, period, and sequence, because that could complicate the analysis.
Crossover is relatively easy when the treatment can be rapidly started and stopped. However, crossover strategy with random allocation of the order of therapy and adequate washout period as discussed earlier is essential for valid results from such self-control designs. Crossover design cannot be adopted if cure or death is the outcome. In addition, regulatory agencies seldom approve crossover because of the possibility of carry-over effect even if demonstrated to be absent. Also, side-effects and tolerability are difficult to study in a crossover setup since the administration is for a short period. Thus, crossover is not recommended for phase-III trials. But this design can be easily used in phase-II, particularly for identifying the right dosage. The conventional crossover is two regimens–two periods–two sequences strategy. But it can be extended for multiple periods and multiple regimens. For further details, see Senn [1]. ... ...
For final and full version, see
Concise Encyclopedia of Biostatistics for Medical Professionals
